Too much insulin can be as dangerous as too little: Research
A little more than a century has gone since the discovery of insulin, and since then hormone's therapeutic abilities have grown and refined. Read further on Dynamite News:
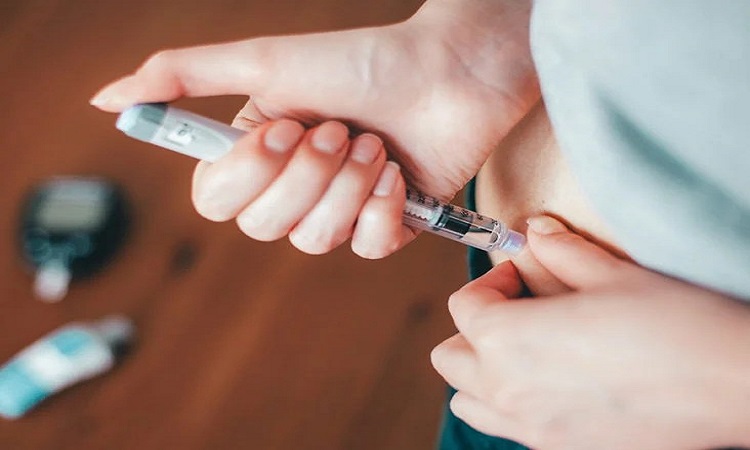
Washington: A little more than a century has gone since the discovery of insulin, and since then hormone's therapeutic abilities have grown and refined. Insulin is a critical treatment for type 1 diabetes and, in many cases, type 2 diabetes. According to the American Diabetes Association, 8.4 million Americans use insulin.
One hundred years of research have greatly advanced medical and biochemical understanding of how insulin works and what happens when it is lacking, but the reverse, how potentially fatal insulin hyper-responsiveness is prevented, has remained a persistent mystery.
In a new study, published in the April 20, 2023 online edition of Cell Metabolism, a team of scientists at the University of California San Diego School of Medicine, with colleagues elsewhere, describe a key player in the defense mechanism that safeguards us against excessive insulin in the body.
"Although insulin is one of the most essential hormones, whose insufficiency can result in death, too much insulin can also be deadly," said senior study author Michael Karin, PhD, Distinguished Professor of Pharmacology and Pathology at UC San Diego School of Medicine.
"While our body finely tunes insulin production, patients who are treated with insulin or drugs that stimulate insulin secretion often experience hypoglycemia, a condition that if gone unrecognized and untreated can result in seizures, coma and even death, which collectively define a condition called insulin shock."
Hypoglycemia (low blood sugar) is a significant cause of death among persons with diabetes.
Also Read |
Want to keep diabetes at bay? Drink alcohol
In the new study, Karin, first author Li Gu, PhD, a postdoctoral scholar in Karin's lab, and colleagues describe "the body's natural defense or safety valve" that reduces the risk of insulin shock.
That valve is a metabolic enzyme called fructose-1,6-bisphosphate phosphatase or FBP1, which acts to control gluconeogenesis, a process in which the liver synthesizes glucose (the primary source of energy used by cells and tissues) during sleep and secretes it to maintain steady supply of glucose in the bloodstream.
Some antidiabetic drugs, such as metformin, inhibit gluconeogenesis but without apparent ill effect. Children born with a rare, genetic disorder in which they do not produce sufficient FBP1 can also remain healthy and live long lives.
But in other cases, when the body is starved for glucose or carbohydrates, an FBP1 deficiency can result in severe hypoglycemia. Without a glucose infusion, convulsions, coma and possibly death can ensue.
Compounding and confounding the problem, FPB1 deficiency combined with glucose starvation produces adverse effects unrelated to gluconeogenesis, such as an enlarged, fatty liver, mild liver damage and elevated blood lipids or fats.
Also Read |
Tackling diabetes amongst pregnant women
To better understand the roles of FBP1, researchers created a mouse model with liver specific FBP1 deficiency, accurately mimicking the human condition. Like FBP1-deficient children, the mice appeared normal and healthy until fasted, which quickly resulted in the severe hypoglycemia and the liver abnormalities and hyperlipidemia described above.
Gu and her colleagues discovered that FBP1 had multiple roles. Beyond playing a part in the conversion of fructose to glucose, FBP1 had a second non-enzymatic but critical function: It inhibited the protein kinase AKT, which is the primary conduit of insulin activity. "Basically, FBP1 keeps AKT in check and guards against insulin hyper-responsiveness, hypoglycemic shock and acute fatty liver disease," said first author Gu.
Working with Yahui Zhu, a vising scientist from Chongqing University in China and second author of the study, Gu developed a peptide (a string of amino acids) derived from FBP1 that disrupted the association of FBP1 with AKT and another protein that inactivates AKT.
"This peptide works like an insulin mimetic, activating AKT," said Karin. "When injected into mice that have been rendered insulin resistant, a highly common pre-diabetic condition, due to prolonged consumption of high-fat diet, the peptide (nicknamed E7) can reverse insulin resistance and restore normal glycemic control."
Karin said the researchers would like to further develop E7 as a clinically useful alternative to insulin "because we have every reason to believe that it is unlikely to cause insulin shock." (ANI)
 Dynamite News
Dynamite News 